Trump Signs Executive Order to Accelerate Psychedelic Drug Reviews, Targeting Treatments for Veterans’ PTSD and TBI
The move fulfills pledges from Health Secretary Robert F. Kennedy Jr. and reflects rare bipartisan interest in expanding access to psychedelics for mental health conditions.
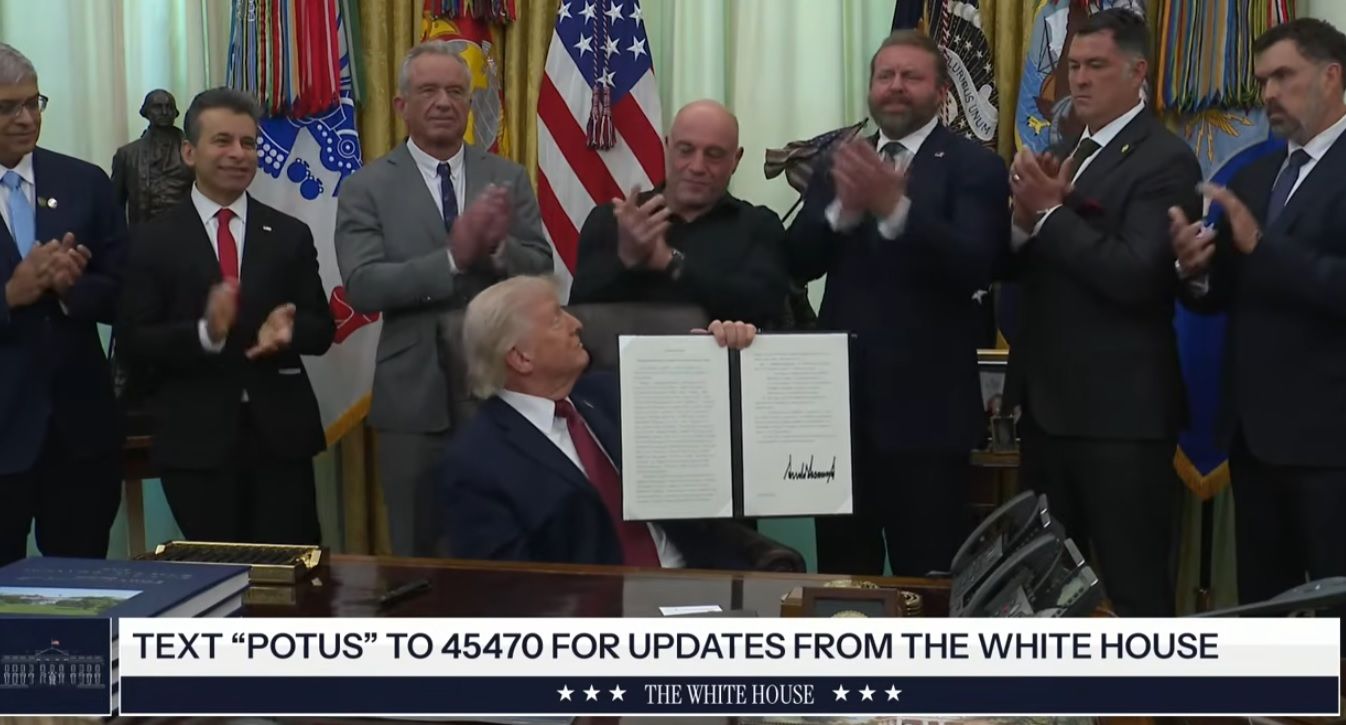
WASHINGTON — President Donald Trump signed an executive order on Saturday directing the Food and Drug Administration to dramatically speed up reviews of certain psychedelic drugs, with a strong focus on ibogaine for treating post-traumatic stress disorder, traumatic brain injury, and opioid addiction in veterans.
“Today’s order will ensure that people suffering from debilitating symptoms might finally have a chance to reclaim their lives and lead a happier life,” Trump said during the Oval Office signing ceremony. He added that the directive will “dramatically accelerate” access to research and treatments, noting, “If these turn out to be as good as people are saying, it’s going to have a tremendous impact.”
The move fulfills pledges from Health Secretary Robert F. Kennedy Jr. and reflects rare bipartisan interest in expanding access to psychedelics for mental health conditions. Trump was joined by top health officials, conservative podcaster Joe Rogan, and retired Navy SEAL Marcus Luttrell, whose story inspired the film “Lone Survivor.” Rogan revealed he had texted Trump information on ibogaine, prompting the president’s quick response: “Sounds great. Do you want FDA approval? Let’s do it.” Luttrell told Trump the treatment “absolutely changed my life for the better” and would “save a lot of lives.”
Ibogaine, derived from a West African shrub and used in traditional Bwiti religious ceremonies, remains a Schedule I controlled substance with no accepted medical use under federal law. It is banned due to risks including irregular heart rhythms and has been linked to more than 30 deaths in medical literature. The National Institutes of Health halted early research in the 1990s over cardiovascular toxicity concerns. Despite these risks, U.S. veterans have reported significant relief from PTSD and TBI symptoms after traveling to clinics in Mexico, where the drug is administered with heart monitoring and emergency equipment.
Next week, the FDA will issue national priority vouchers for three psychedelics already designated as breakthrough therapies — the first time such fast-track status has been extended to any psychedelics. The agency is also clearing the path for the first-ever U.S. human trials of ibogaine. No psychedelic drugs have yet received full FDA approval, though psilocybin, MDMA, and LSD are in large-scale trials for mental health conditions. Oregon and Colorado have legalized psilocybin therapy at the state level.
The order builds on Texas’s 2025 law, backed by veterans groups and former Gov. Rick Perry, which allocated $50 million for ibogaine research. Perry and groups like Americans for Ibogaine have pushed to reduce federal barriers.
For combat veterans and their families struggling with treatment-resistant PTSD and brain injuries after years of war, the executive action offers hope that promising therapies long stalled by federal restrictions could soon reach those who need them most. Clinics like Beond Ibogaine in Cancun, which treated 2,000 patients last year (including 100 veterans at no cost), say the federal acknowledgment marks a shift from “fringe and underground” to legitimate medical research. The order does not reclassify ibogaine or provide immediate insurance coverage, but it signals a major policy pivot under the Trump administration.
Join the Team
Are you trying to break into news writing but struggling to get published at major outlets? At RWT News, we're always looking for talented, motivated writers who share our commitment to straightforward, factual conservative journalism. If you believe in honest reporting and want real experience and bylines, we'd love to hear from you.
Visit our Join the Team page to learn more and contact us directly.
